Read Our Article
Central Venous Catheter Kit: Essential Tools for Safe and Effective Catheter Insertion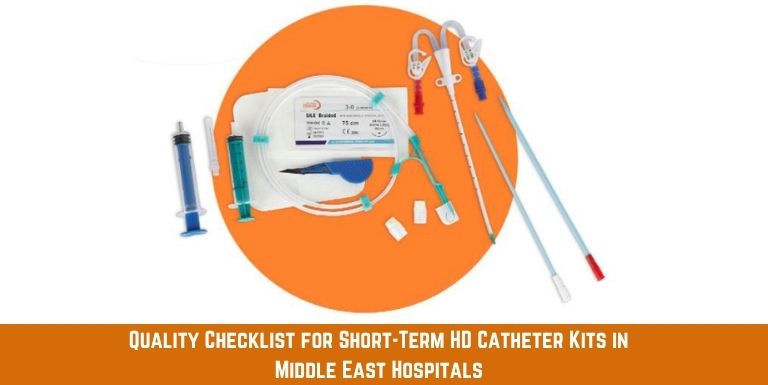
Hospitals across Saudi Arabia, the UAE, Qatar, Kuwait, Bahrain and Oman are facing growing demand for reliable short-term hemodialysis catheter kits as chronic kidney disease prevalence rises in the region. Procurement teams must evaluate product quality, sterility, regulatory compliance and vendor reliability before committing to bulk orders. Working with a relaible short-term hemodialysis catheters manufacturer helps hospitals manage cost without compromising patient safety. A structured quality checklist streamlines the evaluation process by covering material standards, packaging integrity, clinical certifications and post-sale support. This article outlines a practical, step-by-step procurement framework tailored to the regulatory and clinical realities of Middle Eastern healthcare systems, helping procurement officers make informed, compliant and cost-effective sourcing decisions.
Hemodialysis is a life-sustaining treatment for patients with acute or end-stage kidney failure and access to safe vascular access devices is non-negotiable. Across the Middle East, especially in Saudi Arabia, the UAE, Qatar, Kuwait, Bahrain and Oman, hospitals are scaling up dialysis capacity in response to rising rates of diabetes and hypertension-two leading causes of chronic kidney disease. As demand surges, procurement departments face mounting pressure to source reliable short-term dialysis catheter kits quickly and cost-effectively.
Yet speed should never override diligence. A compromised short-term hemodialysis catheter can cause bloodstream infections, thrombosis or catheter malposition-each carrying serious clinical and legal consequences. This article presents a structured quality checklist designed specifically for Middle East hospital buyers, procurement officers and supply chain managers to evaluate products, suppliers and compliance benchmarks before finalizing orders.
A short-term hemodialysis catheter is a dual- or triple-lumen central venous catheter inserted for temporary vascular access-typically used for days to weeks in ICU or acute nephrology settings. Kits include the catheter itself, a guidewire, dilator, introducer needle, syringe and fixation components, all pre-packaged for single use.
Understanding what is included in the kit helps procurement teams compare offerings accurately. Not all kits from every short-term hemodialysis catheters are assembled equally. Some include heparin caps and suture wings; others do not. Buyers must define their clinical requirements first, then match kit specifications to those needs-rather than accepting the default configuration offered by a short-term hemodialysis catheter kit wholesaler.
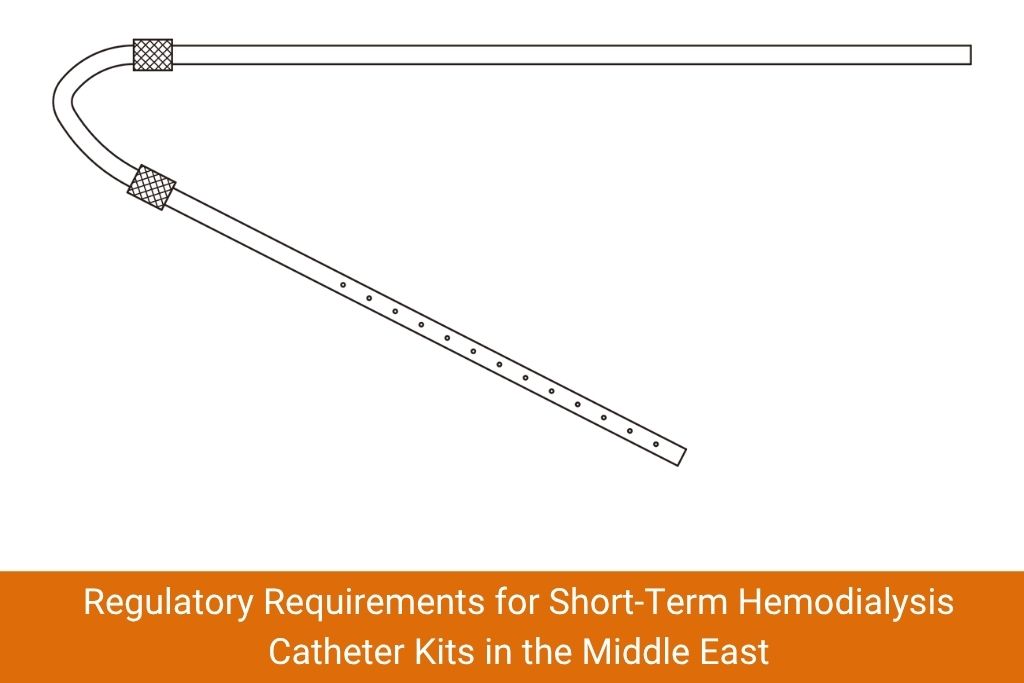
The Saudi Food and Drug Authority mandates that all medical devices, including the short-term dialysis catheter kit, carry valid SFDA registration. Suppliers must provide a Medical Device Marketing Authorization (MDMA) number. Buyers should verify registration status on the SFDA’s online portal before signing any contract.
The UAE requires that devices meet MOHAP or DHA registration depending on the emirate. Products must also conform to ISO 10993 (biocompatibility testing) and ISO 11135 (sterilization). Importers must hold valid import licenses from the respective health authority.
Qatar’s Ministry of Public Health mandates CE marking or FDA clearance for imported medical devices. Procurement teams should confirm that any short-term hemodialysis catheter kit purchased carries traceable certification documentation valid for the Gulf region.
Kuwait, Bahrain and Oman each maintain national health authority registries that require device registration and technical dossiers. Bahrain’s National Health Regulatory Authority (NHRA) in particular has tightened documentation standards, requiring conformity certificates translated to Arabic for local customs clearance.
Before committing to any short-term hemodialysis catheter kit bulk supplier, procurement teams should apply the following product-level criteria:
A product’s quality is inseparable from its supply chain. When assessing a short-term hemodialysis catheter kit wholesaler, apply the following benchmarks:
Our Other Product
Double Lumen CatheterProcurement decisions should not be made in isolation from clinical input. Engage nephrologists, intensivists and infection control teams to validate:
After shortlisting vendors, procurement officers must formalize agreements that protect the hospital’s interests over the contract duration:
Hospitals procuring short-term dialysis catheter kits at volume should consider tendering to multiple pre-qualified suppliers simultaneously to create competitive pricing. However, the lowest bid should never be the only selection criterion.
Consolidating orders through a regional short-term hemodialysis catheter kit wholesaler with GCC distribution capabilities can also reduce per-unit landed costs, customs clearance time and local warehousing overhead-without the risk of working with unverified manufacturers.
Procuring short-term hemodialysis catheter kits for Middle East hospitals requires far more than price negotiation. From SFDA registration in Saudi Arabia to NHRA certification in Bahrain, each country presents distinct regulatory requirements that must be met before any short-term dialysis catheter kit reaches the clinical setting. A rigorous quality checklist covering product materials, sterility, packaging, supplier credentials and contractual safeguards is the most reliable framework for making procurement decisions that protect patients, comply with local law and deliver sustainable value. Hospitals that engage certified short-term hemodialysis catheters manufacturers and vetted wholesalers through structured evaluation processes consistently achieve better clinical outcomes and fewer supply disruptions-making the checklist not just a best practice, but a procurement imperative.
They should hold at minimum ISO 13485 quality management certification, CE marking and ideally FDA 510(k) clearance, alongside valid registration in the relevant Middle East country.
Request hospital client references within the GCC, confirm their import licenses with local health authorities and verify that they provide full regulatory documentation support for each country.
A minimum of 24 months from the manufacturing date is recommended for bulk purchases to allow adequate inventory rotation without risk of expiry before clinical use.
Yes, but the supplier must hold country-specific registrations for Saudi Arabia (SFDA), UAE (MOHAP/DHA), Qatar (MOPH), Kuwait, Bahrain (NHRA) and Oman separately-one registration does not cover all.
Industry standard acceptable quality level (AQL) for sterile medical devices is typically under 0.5% per batch; this should be explicitly stated in procurement contracts with any bulk supplier.

We are recognized as one of the leading manufacturers, exporters, and suppliers of a diverse range of high-quality medical devices in India. Our products, including Interventional Cardiology, Nephrology, Anaesthesia, General Consumables, and Critical Care Accessories, are designed to meet the highest standards of excellence, ensuring reliable performance and patient care.
A – 8, Kuraopokpi industrial Estate,
Thoubal (kakching) Imphal,
Manipur – 795001, India.