Read Our Article
How Spinal Needle Innovation Improves Safety in Epidural and Spinal Anesthesia Procedures?
The shift toward plasticizer-free polymer catheters in epidural catheter kits represents a significant advancement in anesthesia safety and product biocompatibility. Traditional PVC-based epidural catheters use DEHP and other chemical softeners that can leach into spinal tissues, raising concerns about neurotoxicity, hormonal disruption and long-term patient harm. Modern epidural sets now incorporate nylon, polyurethane or PTFE-based catheters that eliminate these risks without sacrificing flexibility or clinical performance. Reputable epidural set manufacturers are responding to regulatory demands and clinical evidence by reformulating their product lines to meet ISO 10993 biocompatibility standards. Clinicians, hospital procurement teams and healthcare systems must prioritize these safer alternatives when evaluating epidural needle and catheter procurement decisions.
Pain management during labor, surgery and post-operative recovery has long depended on regional anesthesia – and at the heart of this practice is the epidural kit. While attention typically focuses on needle gauge, catheter threading technique and filter integration, the polymer composition of the catheter itself has emerged as a critical patient safety consideration. Specifically, the presence or absence of chemical plasticizers in catheter materials directly affects biocompatibility, long-term tissue exposure and overall procedural safety.
This article explores why plasticizer-free polymers are becoming the standard in modern epidural catheter kits, what the clinical evidence says and how procurement decisions made by hospitals and clinicians can either protect or compromise patient outcomes.
Most traditional epidural catheters are manufactured from polyvinyl chloride (PVC), a material that requires chemical additives – plasticizers – to achieve the softness and flexibility necessary for safe threading into the epidural space. The most commonly used plasticizer is DEHP, which makes up as much as 40% of the catheter’s total weight.
The problem is chemical migration. During extended dwell periods – common in labor epidurals that may remain in place for 12 to 24 hours or longer – DEHP and related phthalates can leach from the catheter into the surrounding tissue and cerebrospinal fluid. Studies in toxicology and reproductive medicine have identified phthalates as endocrine disruptors capable of interfering with hormonal signaling, particularly in neonates and pregnant patients who are already physiologically vulnerable.
A complete epidural catheter set typically includes the epidural needle, catheter, filter, connectors and loss-of-resistance syringe. If the catheter component contains migrating plasticizers, every patient receiving epidural analgesia faces a measurable, preventable chemical exposure risk.
Choosing between a Quincke-type and a pencil-point spinal needle often depends on the procedure being performed and the desired outcome. Both types have specific applications that align with their unique features, making them suited for different spinal procedures.
The Quincke-type spinal needle is widely used in diagnostic procedures like lumbar punctures, where the primary goal is to collect cerebrospinal fluid (CSF) for analysis. The sharp bevel of the Quincke needle allows for precise entry into the subarachnoid space, where the CSF resides. The controlled cutting action makes it easier to puncture the dura mater with minimal effort, providing a fast and effective way to access the spinal fluid without causing significant tissue damage. However, while the Quincke needle offers great precision, its sharp bevel can sometimes cause a larger puncture hole, which may increase the risk of post-procedural headaches or a cerebrospinal fluid leak if not properly managed.
The transition to plasticizer-free polymers in the epidural set is not merely a marketing claim – it reflects genuine advances in materials science. The primary alternatives to plasticized PVC include:
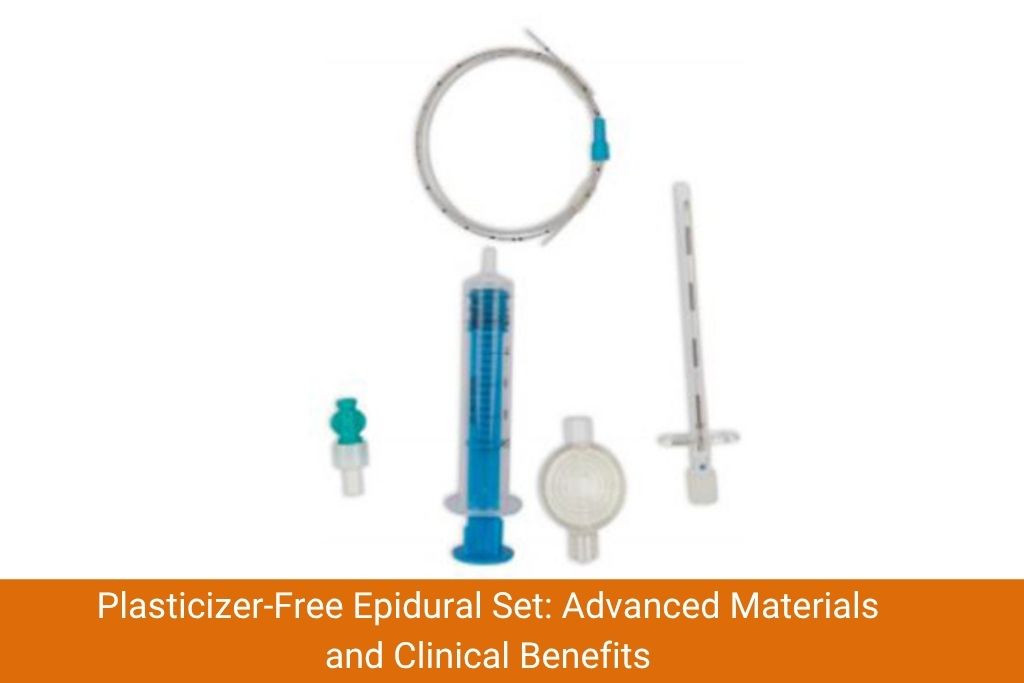
These materials eliminate leachable plasticizer content while maintaining the clinical performance clinicians require. A plasticizer-free epidural set also reduces drug adsorption, meaning the anesthetic agent reaches the patient more predictably – improving dosing accuracy and analgesic consistency.
Our Other Product
Bone Marrow Biopsy NeedleOne of the most important factors when choosing between a Quincke and pencil-point spinal needle is the need for precision. Both types offer a level of precision, but in different ways. The Quincke needle, with its sharp bevel, provides excellent tactile feedback, allowing practitioners to feel resistance and gauge depth during insertion. This feedback is crucial for navigating delicate tissues and ensuring the needle reaches the correct location in the spinal column.
However, the precision offered by the quincke needle may come at the cost of increased tissue damage. Its cutting bevel may cause more trauma to surrounding tissues, particularly in highly sensitive areas like the epidural space. In contrast, the pencil-point needle’s rounded, non-cutting design reduces tissue trauma, allowing for a smoother insertion. The lack of sharp cutting edges means that the needle glides through tissues more gently, but this may also result in less tactile feedback during the procedure.
When conducting spinal procedures that require extreme accuracy, such as spinal anesthesia or the placement of a spinal introducer needle, the precision and tactile feedback of a Quincke-type needle can be more advantageous. Its cutting bevel allows for a more controlled insertion into specific target areas, which is essential when navigating the spinal column.
Regulatory frameworks are increasingly addressing material safety in implantable and body-contact medical devices. ISO 10993 – the international standard governing biological evaluation of medical devices – requires manufacturers to assess cytotoxicity, sensitization and systemic toxicity of all materials that contact the body.
For epidural catheter kits, this means the polymer, adhesives, coatings and any lubricants must be evaluated for leachables and extractables. Devices sold in the European Union under the Medical Device Regulation face stricter justification requirements for phthalate content. In the United States, the FDA has issued guidance on DEHP-containing devices and recommends alternative materials where feasible.
Choosing epidural catheter sets that comply with these evolving standards is not just about regulatory checkboxes – it reflects a genuine commitment to patient-first procurement and harm reduction.
In spinal procedures, minimizing trauma and reducing complications are top priorities. While the Quincke-type spinal needle offers more precision in terms of puncture control, it may cause more tissue damage and increase the likelihood of complications such as headaches or a dural puncture. The pencil-point spinal needle, on the other hand, provides a gentler approach to tissue separation, which helps reduce the risk of bleeding, nerve injury and post-procedural pain.
Both needle types are designed with precision-ground stylets, which help minimize tissue coring and ensure smooth gliding. These features optimize the overall insertion process, reducing friction and improving ease of use. However, when choosing between the two, it’s important to consider the patient’s individual needs and the specific procedure being performed. For example, if the procedure requires a needle with minimal impact on surrounding tissues, a pencil-point needle would likely be the better choice. However, if the focus is on reaching a specific target with precision, a Quincke needle may be more suitable.
The responsibility for safer epidural catheter materials does not rest solely with clinicians. Every epidural set manufacturer must take an active role in eliminating unnecessary chemical risk from their product lines.
Leading manufacturers are now offering DEHP-free and latex-free epidural catheter kits as standard products, investing in nylon and polyurethane catheter formulations that meet or exceed the clinical performance of legacy PVC designs. Some have pursued third-party biocompatibility validation and published extractable/leachable data openly – an important step toward supply chain transparency.
When evaluating an epidural set manufacturer, hospital procurement teams should ask specific questions: What polymer is used in the catheter? Has DEHP been eliminated? Is ISO 10993 testing documentation available? Are extractable compound profiles published? These criteria should form the baseline of any tender specification for epidural kits.
Our Other Product
Cathlab AccessoriesOne of the most important factors when choosing between a Quincke and pencil-point spinal needle is the need for precision. Both types offer a level of precision, but in different ways. The Quincke needle, with its sharp bevel, provides excellent tactile feedback, allowing practitioners to feel resistance and gauge depth during insertion. This feedback is crucial for navigating delicate tissues and ensuring the needle reaches the correct location in the spinal column.
However, the precision offered by the quincke needle may come at the cost of increased tissue damage. Its cutting bevel may cause more trauma to surrounding tissues, particularly in highly sensitive areas like the epidural space. In contrast, the pencil-point needle’s rounded, non-cutting design reduces tissue trauma, allowing for a smoother insertion. The lack of sharp cutting edges means that the needle glides through tissues more gently, but this may also result in less tactile feedback during the procedure.
When conducting spinal procedures that require extreme accuracy, such as spinal anesthesia or the placement of a spinal introducer needle, the precision and tactile feedback of a Quincke-type needle can be more advantageous. Its cutting bevel allows for a more controlled insertion into specific target areas, which is essential when navigating the spinal column.
Plasticizer safety cannot be assessed in isolation. The epidural needle used to access the epidural space sets the stage for catheter introduction and the compatibility between needle design and catheter material matters clinically. A Tuohy needle paired with a stiff, inadequately lubricated catheter increases tissue trauma risk; a well-matched epidural needle and catheter system minimizes resistance during threading and reduces the chance of paresthesia, intravascular placement or catheter shear.
Modern epidural catheter kits are designed with system-level thinking – matched inner diameters, atraumatic catheter tips and soft-tip reinforced designs that work with the needle geometry to optimize placement safety. A plasticizer-free catheter in this context also means the catheter retains consistent mechanical properties throughout the procedure, without softening from thermal changes or interaction with drug vehicles.
In settings where epidural analgesia transitions between care areas – from labor and delivery to recovery or from acute pain management to ICU – the epidural transfer kit plays an important role. Transfer kits typically include sterile extension sets, antibacterial filters and in some configurations, replacement catheters.
If any component in the epidural transfer kit contains plasticized PVC, the cumulative patient exposure to leachables increases every time a connection is made or a component is replaced. A fully plasticizer-free transfer kit – from catheter to filter housing to extension tubing – closes this exposure pathway completely.
Healthcare systems committed to comprehensive patient safety should audit all components in their epidural pathway, not just the primary catheter and source transfer kit components from manufacturers who document material composition for every element.
Plasticizer-free polymer catheters represent a clear and achievable improvement in the safety profile of the epidural kit. The clinical evidence supporting phthalate elimination is compelling, the alternative materials are proven and the regulatory momentum is firmly in the direction of phased removal of DEHP from patient-contact devices. For clinicians, the choice of an epidural catheter set should now include biocompatibility criteria alongside traditional performance metrics. For procurement teams, selecting an epidural set manufacturer that proactively publishes material data and meets current biocompatibility standards is both a patient safety imperative and a defensible institutional standard of care. The epidural catheter itself is a small component in a complex procedure – but its chemistry matters far more than its size suggests.
Plasticizer-free epidural catheter kits eliminate leachable chemicals like DEHP that can migrate into spinal tissue, reducing the risk of endocrine disruption and neurotoxic exposure during extended catheter dwell times.
Not all epidural sets are plasticizer-free; procurement teams should specifically request ISO 10993-compliant, DEHP-free documentation from the epidural set manufacturer before purchasing.
Yes - plasticized PVC can adsorb some anesthetic agents, whereas plasticizer-free catheters made from nylon or polyurethane deliver drugs more predictably and consistently to the patient.
The epidural needle and catheter must be dimensionally compatible; a well-matched system reduces placement trauma and ensures the catheter's mechanical integrity is maintained throughout the procedure.
Absolutely - every component in the epidural transfer kit, including filters and extension tubing, should be assessed for PVC and phthalate content to prevent cumulative chemical exposure during patient transfers.

We are recognized as one of the leading manufacturers, exporters, and suppliers of a diverse range of high-quality medical devices in India. Our products, including Interventional Cardiology, Nephrology, Anaesthesia, General Consumables, and Critical Care Accessories, are designed to meet the highest standards of excellence, ensuring reliable performance and patient care.
A – 8, Kuraopokpi industrial Estate,
Thoubal (kakching) Imphal,
Manipur – 795001, India.