Pediatric interventional procedures demand precision tools that can navigate smaller, more delicate anatomical structures without causing trauma. Among these tools, hydrophilic guidewires have become indispensable – yet their use in children presents distinct challenges that differ significantly from adult applications. From neonatal catheterizations to pediatric urology and cardiology, understanding how these wires perform in young patients is critical for clinical outcomes.
Hydrophilic coated guidewire technology relies on a polymer surface that becomes extremely slippery when wet, reducing friction as it navigates through vessels and ducts. In pediatric patients, vessel diameters can be as small as 1–2 mm in neonates, requiring wires that are not only ultra-thin but also highly responsive to torque. Standard adult-sized wires – even coated ones – can buckle, kink or fail to transmit rotational force accurately in such narrow pathways.
The hydrophilic coating itself introduces a layer of complexity in children. Because the coating reduces friction so dramatically, there is a heightened risk of the guidewire advancing too quickly or too far before the clinician can react – a concern amplified in fragile pediatric vasculature. Selecting the right wire diameter, tip stiffness and coating length becomes a precise clinical decision rather than a standard protocol.
Hydrophilic guidewire performance in pediatric cases is particularly tested in tortuous anatomy. Congenital anomalies, narrowed bile ducts in biliary atresia or stenosed renal collecting systems in children often present geometries that challenge even experienced interventionalists. The wire must be soft enough to follow bends without perforating tissue, yet stiff enough to provide support for catheter advancement.
In pediatric endourology, for instance, access through the urethra to the bladder or ureter in infants requires wires that can make tight turns without losing shape integrity. Hydrophilic wires with floppy distal tips and gradually increasing core stiffness toward the proximal end are often preferred. However, this design must be balanced against the risk of the wire coiling within a small-capacity structure – a problem less common in adults.
Similarly, in pediatric interventional cardiology, navigating congenital heart defects such as patent ductus arteriosus or pulmonary artery stenosis requires hydrophilic guidewires capable of precise directional control through unusual anatomical configurations not seen in mature cardiovascular systems.
Hydrophilic guidewires are widely used in pediatric urological procedures including ureteral stent placement, percutaneous nephrolithotomy access in children and management of ureteropelvic junction obstruction. The challenge here is not just size – it is the combination of a narrow working space, a moving target (especially in awake or lightly sedated children) and the need to maintain sterile access without repeated manipulation.
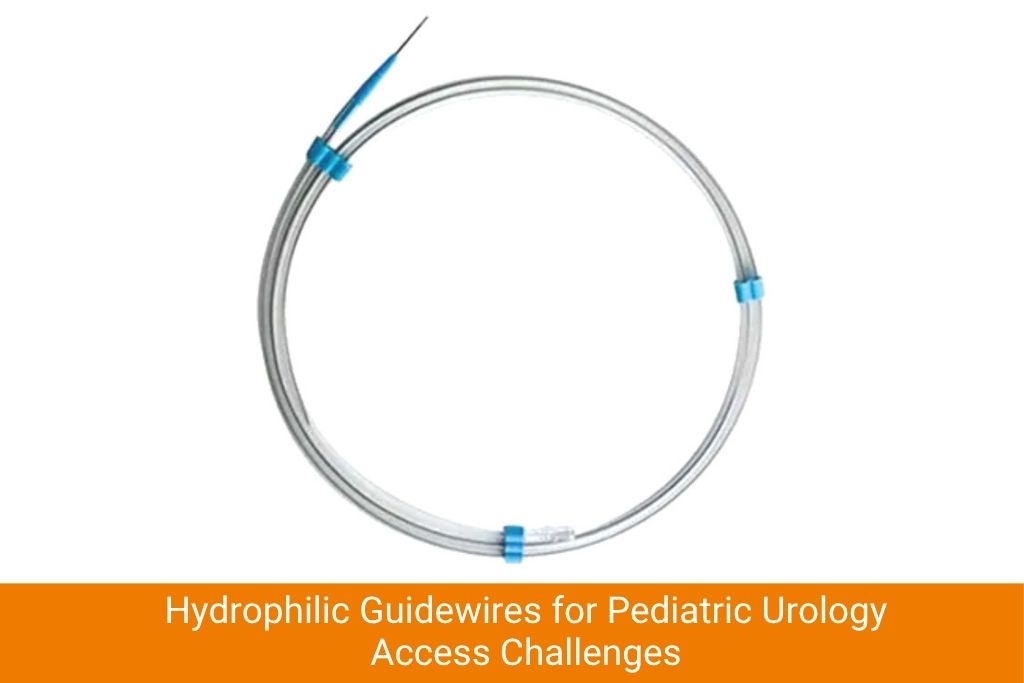
Pediatric urologists frequently report that standard hydrophilic wire lengths, designed for adult procedural tables and fluoroscopy setups, are unnecessarily long for pediatric cases. Excess wire length creates loop management issues outside the body, increasing the risk of contamination or accidental dislodgement. Purpose-designed pediatric lengths – typically 60–100 cm rather than the adult standard of 145–180 cm – would improve workflow considerably.
Additionally, tip load and flexibility grading must be reconsidered for pediatric urological access. A wire that is appropriately flexible for an adult renal pelvis may still be too stiff for an infant’s collecting system, increasing perforation risk during initial access attempts.
Hydrophilic guidewire manufacturer development pipelines are increasingly being pushed by clinical demand to address gaps in pediatric tooling. Historically, the medical device industry produced pediatric equipment as scaled-down versions of adult tools – a method that often fails to account for physiological and anatomical differences.
Forward-thinking manufacturers are now investing in purpose-built pediatric hydrophilic wires that feature thinner cores (as low as 0.014 inches), shorter working lengths, softer distal segments and coating formulations that maintain consistent lubricity across smaller surface areas. Some manufacturers are also exploring dual-layer coatings that offer controlled lubricity – reducing the risk of unintended advancement while still enabling smooth navigation.
Steerable tip technology, already available in some adult neurovascular wires, is beginning to appear in pediatric-sized hydrophilic guidewire prototypes. This innovation could significantly reduce the number of wire exchanges needed during complex pediatric interventions, decreasing procedure time and radiation exposure – both of which are particularly important in children.
Hydrophilic coated guidewire manufacturer compliance with pediatric-specific regulatory guidance remains an evolving area. Regulatory bodies such as the FDA and CE marking authorities in Europe have raised expectations around pediatric device labeling, clinical evidence for pediatric indications and age-specific performance validation.
Many hydrophilic wires currently on the market carry only adult-use indications, with pediatric use falling under off-label clinical judgment. This creates medicolegal uncertainty for interventional radiologists, pediatric surgeons and urologists who rely on these tools daily. Greater investment in pediatric clinical trials and post-market surveillance specific to younger patient populations would help close this gap and give clinicians stronger evidential ground for device selection.
Biocompatibility testing for the hydrophilic polymer coating also needs pediatric-specific assessment. Children – particularly neonates and infants – may have different systemic responses to coating shedding or polymer exposure compared to adults. Long-term follow-up data on coating integrity and particulate generation in pediatric applications remains limited.
Hydrophilic guidewire supplier availability for pediatric-specific sizes and configurations remains inconsistent across global markets. In high-income settings, pediatric specialty centers often have access to a broader range of wire calibers and configurations, but procurement is still fragmented – requiring relationships with multiple suppliers to cover the full spectrum of pediatric procedural needs.
In low- and middle-income countries, where pediatric interventional capacity is growing rapidly, access to appropriate hydrophilic guidewire sizes is a significant barrier. Suppliers frequently prioritize high-volume adult product lines, leaving pediatric practitioners to work with whatever adult inventory is available. Standardizing a pediatric-oriented product catalogue across major hydrophilic guidewires suppliers would directly improve patient safety and procedural efficiency in these settings.
Hospital procurement teams often lack the clinical expertise to evaluate guidewire specifications at the pediatric subspecialty level. Closer collaboration between hydrophilic guidewire suppliers, clinical procurement specialists and pediatric interventionalists is needed to ensure that purchasing decisions reflect actual clinical requirements rather than cost-per-unit metrics alone.
The use of hydrophilic guidewires in neonatal patients represents the most demanding application within pediatric medicine. Neonatal vessels and ducts are extraordinarily delicate and the margin for error is minimal. In procedures such as neonatal biliary interventions, hepatic arterial access for metabolic disease management or renal access in cases of severe obstructive uropathy, standard hydrophilic wires – even pediatric versions – may still lack the nuanced stiffness profiling required.
Emerging wire designs incorporating nitinol cores with progressive stiffness transitions, combined with next-generation hydrophilic coatings that resist delamination under repeated flexion, are being studied in preclinical and early clinical settings. These advances hold promise for improving access success rates in neonatal cases while reducing vessel wall trauma.
The incorporation of ultrasound-visible markers into hydrophilic guidewires is another area of development particularly relevant for neonates, where minimizing radiation exposure through fluoroscopy avoidance is a clinical priority. Real-time ultrasound guidance combined with wire tracking capability could redefine neonatal vascular and urological access protocols.
Pediatric interventional medicine places uniquely demanding requirements on hydrophilic guidewire technology – requirements that current product lines only partially address. From neonatal vascular access to pediatric urological and cardiac procedures, the need for purpose-built wire designs, clearer regulatory frameworks and more responsive supplier ecosystems is evident. As clinical demand grows and manufacturers invest in pediatric-specific innovation, hydrophilic guidewires will continue to evolve as safer, more effective tools for the youngest and most vulnerable patient populations.

We are recognized as one of the leading manufacturers, exporters, and suppliers of a diverse range of high-quality medical devices in India. Our products, including Interventional Cardiology, Nephrology, Anaesthesia, General Consumables, and Critical Care Accessories, are designed to meet the highest standards of excellence, ensuring reliable performance and patient care.
A – 8, Kuraopokpi industrial Estate,
Thoubal (kakching) Imphal,
Manipur – 795001, India.