
Epidural anesthesia plays a significant role in pain management, particularly for patients undergoing surgery or experiencing chronic pain. Epidural sets, including the epidural catheter kit, epidural transfer kit and epidural set, are essential components in this procedure. These sets enable medical professionals to deliver anesthesia directly into the epidural space, ensuring effective pain relief for patients during and after surgery.
The critical nature of these devices, their manufacturing standards and quality control processes are paramount in ensuring patient safety and treatment effectiveness. Medical professionals and patients alike rely on epidural sets to function flawlessly, which is why manufacturers and distributors must adhere to stringent quality control measures during the production process. This article will delve into the manufacturing standards and quality control processes that are critical to the production of epidural sets and how they ensure the safety and efficacy of these vital medical devices.
Compliance with Regulatory Standards
Epidural sets must meet rigorous regulatory standards to ensure their safety and effectiveness. These regulations are often dictated by medical device regulatory bodies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA) and other national or international authorities. Manufacturers are required to meet these standards before their products are approved for clinical use.
Compliance with these regulations involves ensuring that the materials used in the construction of the epidural set are biocompatible, durable and suitable for medical procedures. Manufacturers must also conduct thorough testing on the final product to ensure it meets specific performance standards related to functionality, safety and reliability.
Material Selection and Biocompatibility
The materials used in epidural sets must be carefully selected to ensure that they are compatible with human tissues and do not cause adverse reactions. For example, epidural catheter kits often use unique, plasticizer-free polymers that offer excellent biocompatibility. This ensures that the catheter can remain in the body for extended periods without causing irritation, inflammation or other complications.
Additionally, the materials must be flexible yet strong, allowing for easy insertion while also minimizing the risk of injury to the patient. The epidural set must also be transparent or have a radio-opaque line, allowing healthcare professionals to visually monitor the placement of the catheter and ensure it is positioned correctly within the epidural space.
Sterilization and Packaging
Sterilization is an essential aspect of manufacturing epidural sets. These medical devices must be free from contaminants that could cause infections or other complications when introduced into the human body. Manufacturers utilize various sterilization techniques, such as ethylene oxide (EO) sterilization, gamma radiation or autoclaving, to ensure that each epidural set is free from pathogens.
The packaging is also critical to maintaining their sterility. Proper packaging ensures that the device remains sterile until it is ready for use in a clinical setting. Packaging must be robust and secure to prevent any damage to the device during transportation and storage, ensuring that it is ready for immediate use when needed.
Inspection and Testing of Materials
One of the primary steps in the quality control process is the inspection and testing of raw materials. Every material used in the production of epidural sets, from the catheter to the needle and connectors, undergoes strict quality assurance procedures to ensure it meets the necessary specifications. Materials must be free of defects, contamination and inconsistencies that could affect the performance of the final product.
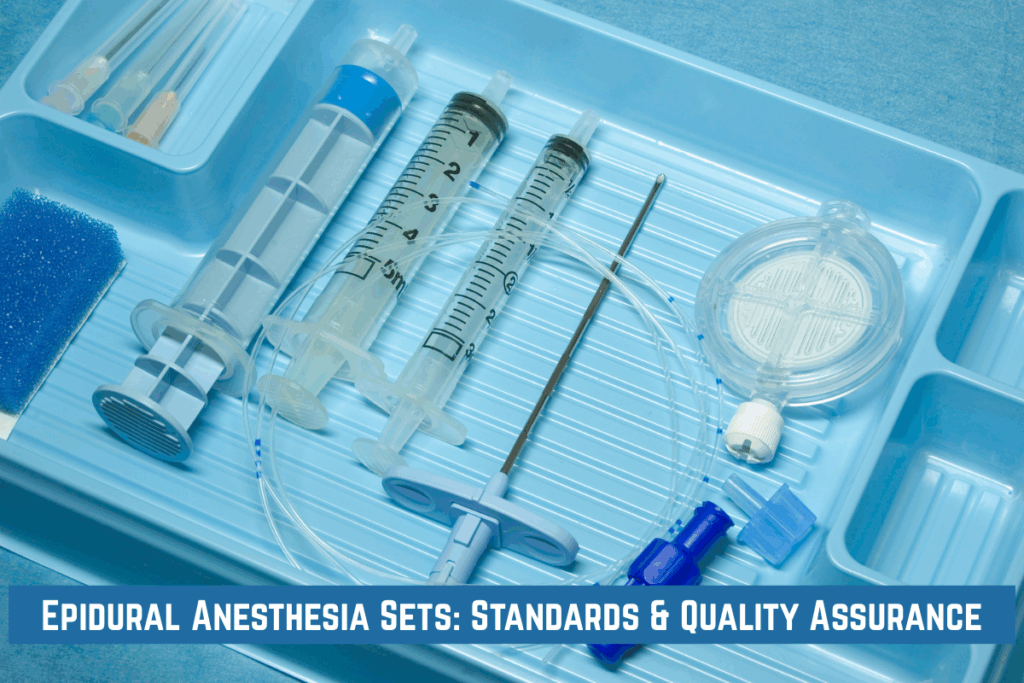
Manufacturers perform tests to ensure that the materials are durable, flexible and suitable for use in a medical environment. Epidural catheter kits must be tested for their strength, flexibility and resistance to kinking, as these factors can influence the ease of catheter insertion and its effectiveness during use.
In-Process Testing During Manufacturing
Throughout the manufacturing process, quality control teams conduct in-process testing to verify that each step of production adheres to established standards. This might include testing the dimensions of the epidural catheter to ensure it meets the required specifications, verifying that the catheter’s soft tip is atraumatic and designed for minimal tissue damage and ensuring that the threading assist guide functions as intended.
In-process testing also ensures that the Epidural Set components, such as the luer lock twist connector and hydrophobic filters, are correctly assembled and meet the quality standards. Any deviation from the specifications can lead to failure in the performance of the epidural set, which could compromise patient safety and treatment efficacy.
End-of-Line Testing and Final Inspections
Once the epidural sets are fully assembled, they undergo a series of final inspections to ensure that the product is ready for use. This includes testing the functionality of the twist clamp on the epidural transfer kit, ensuring the smooth operation of the detachable connector and verifying the overall integrity of the device.
Manufacturers also perform sterility testing to ensure that the epidural catheter set is free from bacterial or fungal contamination. Final inspections include visual checks for defects, leaks or other issues that could compromise the performance of the epidural set.
Packaging and Labeling Control
After the epidural sets have passed all quality control tests, they are carefully packaged in a sterile environment to maintain their sterility. The packaging is labeled with important information, including product details, manufacturing lot number, expiration date and sterilization method. This ensures traceability and provides healthcare providers with the information they need to use the device properly.
Quality control also includes ensuring that packaging is secure, tamper-evident and durable enough to withstand handling and transportation. The epidural set distributor plays a crucial role in ensuring that these packaged devices reach the clinical setting without any compromise to their sterility or functionality.
Epidural set manufacturers are responsible for ensuring that the products they produce meet or exceed regulatory standards and quality control protocols. As part of this responsibility, manufacturers must invest in ongoing research and development to improve the design and functionality of epidural sets and keep up with advancements in medical technology.
Distributors, on the other hand, play a key role in ensuring that the final product reaches healthcare providers in optimal condition. They must ensure proper storage, transport and handling of the epidural sets to maintain their integrity. A strong partnership between manufacturers and distributors is crucial for the successful delivery of safe and effective epidural sets.
The manufacturing standards and quality control processes for Epidural sets are essential to ensuring that these critical medical devices perform as expected and provide the necessary pain relief during procedures. Stringent regulatory compliance, careful material selection, comprehensive testing and thorough inspections all play an integral role in guaranteeing that epidural sets are safe, effective and reliable for patient care.
Manufacturers must adhere to high standards in every phase of the production process, from material sourcing to packaging. Likewise, quality control teams must be vigilant at every stage to prevent defects and ensure that each epidural set meets or exceeds industry expectations. Through collaboration between manufacturers, distributors and healthcare providers, the quality of epidural sets can be maintained, ensuring that these devices continue to deliver safe and effective results in patient care.
By upholding stringent manufacturing standards and rigorous quality control practices, manufacturers and distributors contribute as an essential pain management solution in medical treatments.

We are recognized as one of the leading manufacturers, exporters, and suppliers of a diverse range of high-quality medical devices in India. Our products, including Interventional Cardiology, Nephrology, Anaesthesia, General Consumables, and Critical Care Accessories, are designed to meet the highest standards of excellence, ensuring reliable performance and patient care.
A – 8, Kuraopokpi industrial Estate,
Thoubal (kakching) Imphal,
Manipur – 795001, India.