Read Our Article
How Spinal Needle Innovation Improves Safety in Epidural and Spinal Anesthesia Procedures?
India has grown into a globally recognised hub for surgical and medical disposable products, supplying hospitals domestically and export markets across different countries. Leading medical device manufacturers in India produce a wide range of disposable medical accessories – from gloves and syringes to advanced critical care products – at competitive costs without compromising quality. These products comply with international standards such as ISO, CE and FDA, making Indian manufacturers preferred partners for global procurement. The government’s push through schemes like Make in India and PLI for medical devices has further accelerated capacity and innovation. With growing demand from both public and private healthcare systems, the medical disposables sector in India is positioned for sustained double-digit growth in the coming years.
India today ranks among the top manufacturers and exporters of surgical disposable products in the world. From small district hospitals to large multi-speciality chains, healthcare providers rely heavily on medical disposable products to deliver safe, hygienic and efficient patient care. At the same time, international buyers – particularly from Africa, Southeast Asia, the Middle East and Latin America – are increasingly sourcing from Indian medical device manufacturers because of the strong mix of quality, cost-efficiency and regulatory compliance.
The surgical disposables segment covers everything used once and discarded – products designed to prevent cross-contamination, reduce infection risk and simplify clinical workflows. What makes India stand out is the sheer scale of its manufacturing infrastructure, skilled workforce and the ability to meet international certifications including ISO 13485, CE marking and US FDA registration.
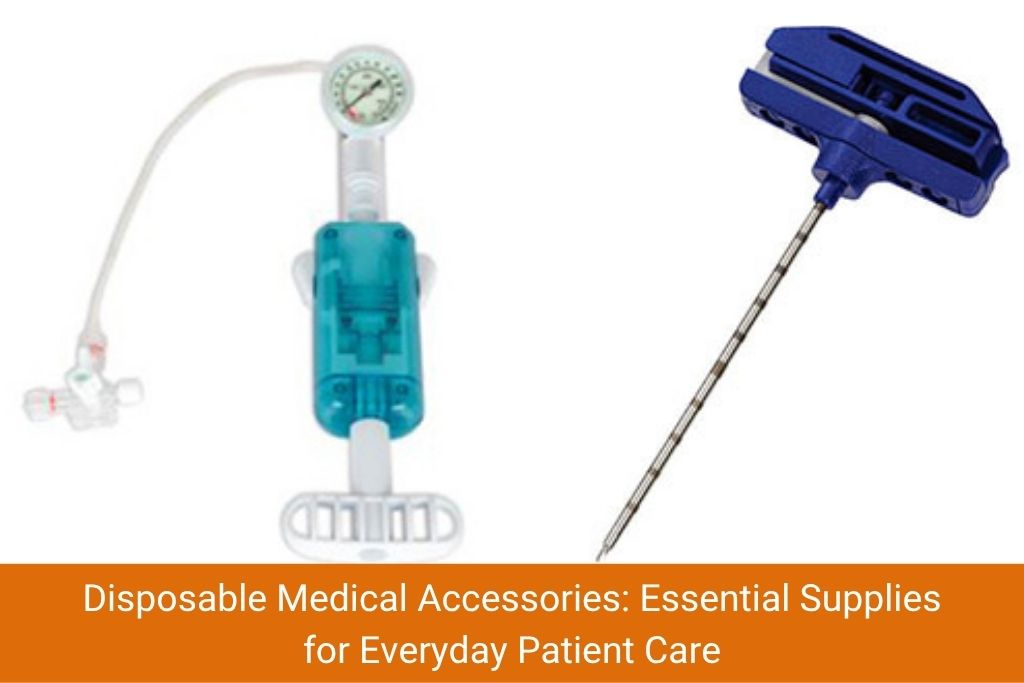
Disposable medical accessories form the backbone of daily clinical operations. These are products every nurse, doctor and paramedic uses multiple times a day – and their quality directly impacts patient safety.
Key disposable medical accessories manufactured in India include:
Indian manufacturers produce these items in large volumes with strict quality controls, making them suitable for both government procurement schemes (like PM-ABHIM and NHM) and private hospital supply chains. Many units operating in Himachal Pradesh, Gujarat and Rajasthan are approved by the Central Drugs Standard Control Organisation (CDSCO) and hold international certifications.
India’s export of medical disposable products has grown significantly over the past decade. According to industry data, India exports medical devices and disposables to over 100 countries, with the US, Germany, Australia and UAE among the top buyers.
High-demand disposable products in the export segment include:
What helps Indian exporters win global contracts is the ability to customise packaging, labelling and product configuration as per buyer specifications. The government’s Medical Devices Rules 2017 brought greater regulatory clarity and this has helped exporters demonstrate compliance more effectively to international procurement agencies.
Choosing between a Quincke-type and a pencil-point spinal needle often depends on the procedure being performed and the desired outcome. Both types have specific applications that align with their unique features, making them suited for different spinal procedures.
The Quincke-type spinal needle is widely used in diagnostic procedures like lumbar punctures, where the primary goal is to collect cerebrospinal fluid (CSF) for analysis. The sharp bevel of the Quincke needle allows for precise entry into the subarachnoid space, where the CSF resides. The controlled cutting action makes it easier to puncture the dura mater with minimal effort, providing a fast and effective way to access the spinal fluid without causing significant tissue damage. However, while the Quincke needle offers great precision, its sharp bevel can sometimes cause a larger puncture hole, which may increase the risk of post-procedural headaches or a cerebrospinal fluid leak if not properly managed.
General & disposables accessories cover a wide spectrum – products used across OPD, emergency, ICU, OT and ward settings. Unlike specialised surgical items, these are required in bulk by virtually every department.
Commonly used general & disposable accessories made in India:
Hospitals and clinics typically procure these through approved vendors tied to central medical stores or through group purchasing organisations (GPOs). Indian manufacturers serving this segment compete primarily on unit price, delivery reliability and shelf-life compliance – all three areas where established Indian suppliers have built strong track records.
Our Other Product
Bone Marrow Biopsy NeedleCritical care products are among the most technically demanding in the disposables space. These items are used in life-sustaining and monitoring roles – errors in quality can have serious clinical consequences. Indian manufacturers have invested heavily in this segment, particularly after the COVID-19 pandemic highlighted gaps in domestic critical care capacity.
Critical care products manufactured and exported from India include:
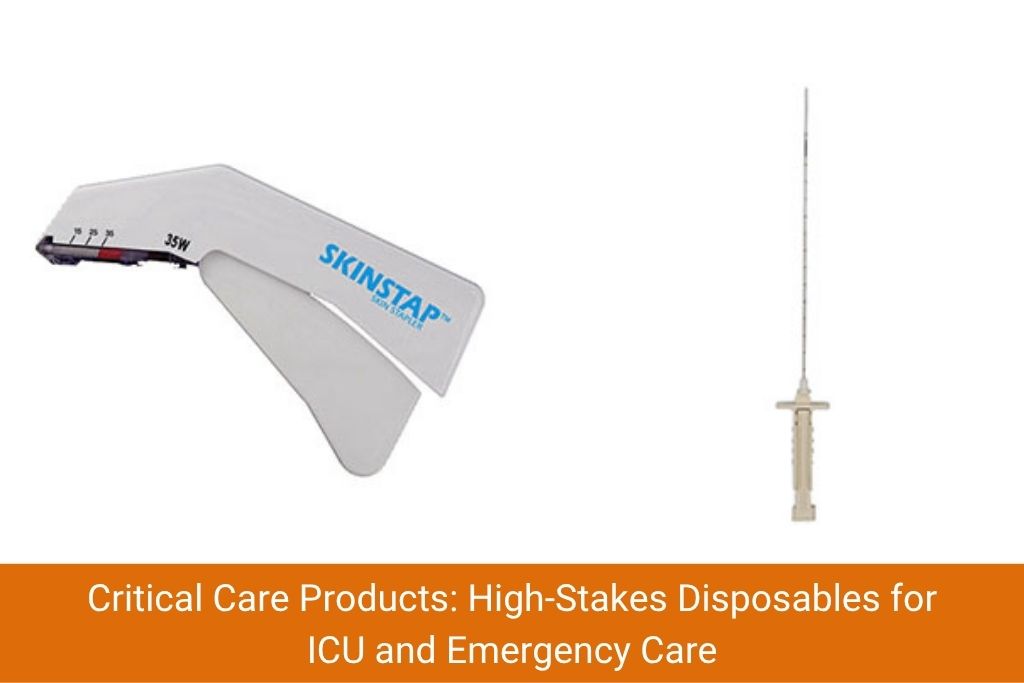
The quality bar for critical care products is significantly higher. Products must go through biocompatibility testing, sterility assurance level (SAL) validation and often require specific regulatory clearances in destination countries.
A credible medical device manufacturer in India today operates under a robust regulatory framework. The Drugs and Cosmetics Act (as amended) and the Medical Devices Rules 2017 define how manufacturers must design, test, produce and track their products.
Key compliance markers that distinguish top manufacturers:
India’s cost advantage is not just about lower labour costs. Raw material availability (rubber, polymers, non-woven fabrics), a large engineering talent pool and clustering of manufacturers in hubs create supply chain efficiencies that are hard to replicate. For hospital procurement teams and international buyers, this translates to competitive pricing without sacrificing compliance standards.
In spinal procedures, minimizing trauma and reducing complications are top priorities. While the Quincke-type spinal needle offers more precision in terms of puncture control, it may cause more tissue damage and increase the likelihood of complications such as headaches or a dural puncture. The pencil-point spinal needle, on the other hand, provides a gentler approach to tissue separation, which helps reduce the risk of bleeding, nerve injury and post-procedural pain.
Both needle types are designed with precision-ground stylets, which help minimize tissue coring and ensure smooth gliding. These features optimize the overall insertion process, reducing friction and improving ease of use. However, when choosing between the two, it’s important to consider the patient’s individual needs and the specific procedure being performed. For example, if the procedure requires a needle with minimal impact on surrounding tissues, a pencil-point needle would likely be the better choice. However, if the focus is on reaching a specific target with precision, a Quincke needle may be more suitable.
Global buyers sourcing disposable medical accessories from India prioritise the following:
India’s disposables manufacturers have worked to strengthen all five areas. Industry bodies like FICCI’s health wing have been instrumental in helping manufacturers upgrade their facilities and navigate export compliance. Many manufacturers also participate in international trade fairs like Medica (Germany), Arab Health (UAE) and Hospitalar (Brazil) to build direct relationships with overseas buyers.
India’s surgical disposables manufacturing sector has matured from a low-cost supplier to a quality-driven, compliance-focused industry. Whether it’s everyday disposable medical accessories or specialised critical care products, Indian medical device manufacturers are capable of meeting the standards demanded by hospitals and healthcare systems worldwide. The combination of regulatory upgrades, government support and increasing private investment in advanced manufacturing is creating a solid foundation for sustained growth. For hospital procurement teams and international distributors, India offers a reliable, scalable and cost-effective source for the full spectrum of medical disposable products. As global healthcare demand continues to rise, India is well-positioned to play an even larger role in the world’s medical supply chain.
India commonly exports surgical gloves, IV sets, syringes, catheters and wound care products to hospitals and distributors in over multiple countries.
Top Indian manufacturers maintain ISO 13485, CE and FDA certifications and operate CDSCO-licensed cleanroom facilities with validated sterilisation and quality control processes.
Yes - Indian-made critical care products like ventilator circuits, CVCs and endotracheal tubes meet international biocompatibility and sterility standards and are actively used in global hospitals.
Indian manufacturers supply examination gloves, surgical drapes, disposable aprons, specimen bags, sterile swabs and procedure kits in bulk to hospital chains and procurement agencies.
India's advantage lies in its combination of low production costs, abundant raw materials, skilled workforce, regulatory compliance infrastructure and growing cluster-based manufacturing hubs.

We are recognized as one of the leading manufacturers, exporters, and suppliers of a diverse range of high-quality medical devices in India. Our products, including Interventional Cardiology, Nephrology, Anaesthesia, General Consumables, and Critical Care Accessories, are designed to meet the highest standards of excellence, ensuring reliable performance and patient care.
A – 8, Kuraopokpi industrial Estate,
Thoubal (kakching) Imphal,
Manipur – 795001, India.