
Temporary vascular access is a critical intervention in acute kidney injury, fluid overload and emergency dialysis settings. A short-term hemodialysis catheter is one of the most commonly placed devices in nephrology and critical care units – yet it carries a significant risk of complications when not handled with clinical precision. From insertion errors to post-placement infections, complications associated with a short-term dialysis catheter kit can lead to prolonged hospitalization, bloodstream infections and vascular damage. This guide provides evidence-based, practical tips for medical staff to minimize those risks at every stage of catheter use.
Short-term hemodialysis catheters are typically non-tunneled, dual-lumen devices inserted into central veins such as the internal jugular, subclavian or femoral vein. Unlike tunneled catheters designed for long-term use, these devices are intended for acute, short-duration access – generally less than three weeks. Because they sit in high-flow central vessels without a subcutaneous tunnel barrier, they are more vulnerable to catheter-related bloodstream infections (CRBSIs), thrombosis and mechanical complications.
The most common complications include:
Understanding these risks is the first step. Preventing them requires discipline across three phases: pre-insertion preparation, intra-procedural technique and post-placement management.
Every short-term dialysis catheter kit includes the essential components – catheter, introducer needle, guidewire, dilator and fixation device. However, the quality of the kit and how staff prepares before use are both equally important.
Site Selection: The right jugular vein is the preferred insertion site for most patients due to its lower risk of stenosis and pneumothorax compared to the subclavian approach. The femoral site should be reserved for patients in whom neck access is contraindicated, as femoral catheters carry a higher infection risk and limit patient mobility.
Ultrasound-Guided Insertion: Real-time ultrasound guidance during needle placement significantly reduces arterial puncture rates and failed cannulation attempts. Clinical guidelines from KDIGO and NICE strongly recommend ultrasound guidance as the standard of care for central venous catheter insertion. Medical staff should be trained and competent in ultrasound-assisted technique before independently placing these devices.
Sterile Barrier Precautions: The procedure must be performed under maximal sterile barrier (MSB) precautions – full sterile draping, sterile gown, gloves, cap and mask. Evidence consistently shows that MSB reduces CLABSI rates. Even in emergency settings, these precautions should not be bypassed.
Skin Antisepsis: Chlorhexidine-based solutions are the recommended antiseptic for skin preparation before insertion. Allow the solution to fully dry before proceeding with needle puncture to achieve maximum antimicrobial effect.
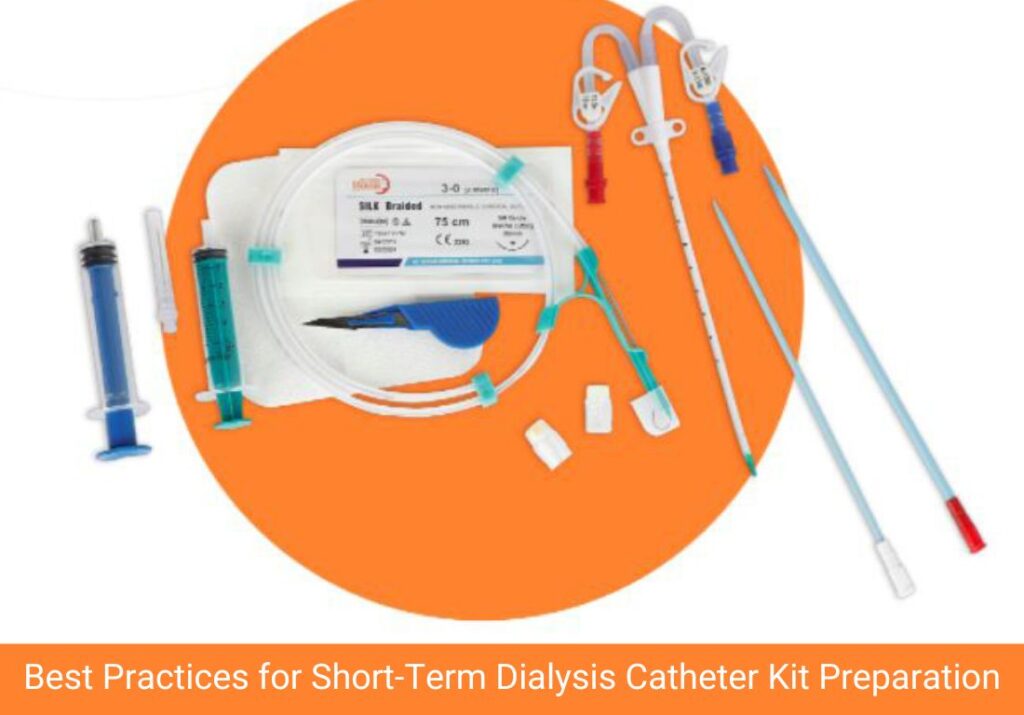
During placement of short-term hemodialysis catheters, technical precision directly determines the rate of mechanical complications.
Seldinger Technique and Wire Management: The modified Seldinger technique – needle access, guidewire insertion, dilation and catheter advancement – is standard. One of the most preventable errors is advancing the guidewire against resistance, which risks vessel perforation or cardiac arrhythmia if the wire enters the right atrium. Always confirm wire position with fluoroscopy or ECG-guided placement when available.
Catheter Length Selection: Incorrect catheter length leads to malposition. For right internal jugular access, the catheter tip should sit at the cavoatrial junction. Inserting too deep risks intracardiac placement; too shallow compromises flow rates during hemodialysis sessions.
Post-Insertion Confirmation: A chest X-ray is mandatory after internal jugular or subclavian placement to confirm catheter tip position and rule out pneumothorax before initiating dialysis. Skipping this step is a patient safety violation.
Secure Fixation: Use the suture wings or securement device included in the short-term dialysis catheter kit to anchor the catheter firmly at the exit site. Movement at the insertion point increases the risk of site trauma, contamination and catheter dislodgement.
Infection is the leading complication associated with any short-term hemodialysis catheter and most CRBSIs are preventable with consistent post-placement care.
Daily Exit-Site Assessment: Nursing staff should inspect the catheter exit site at every shift – looking for redness, swelling, purulent discharge or tenderness. Any signs of local infection must be escalated promptly and catheter removal should be considered without delay.
Dressing Changes: Sterile transparent semipermeable dressings should be changed every 5–7 days or immediately when wet, soiled or loose. Gauze dressings require more frequent changes – every 48 hours. During any dressing change, strict hand hygiene and aseptic non-touch technique (ANTT) must be observed.
Hub Disinfection before Every Access: Before connecting or disconnecting dialysis lines, staff must scrub the catheter hub and needleless connector with a 70% isopropyl alcohol pad for a minimum of 15 seconds and allow it to dry. This “scrub the hub” protocol is the simplest and most effective measure against intraluminal contamination.
Antibiotic Lock Therapy: For patients at high risk of catheter-associated infection or with recurrent CLABSI, antibiotic or antimicrobial lock solutions can be instilled into catheter lumens between dialysis sessions. This reduces bacterial biofilm formation inside the lumen.
Heparin Locking for Patency: After each dialysis session, lumen patency is maintained by instilling heparin solution. The appropriate concentration and volume depend on the catheter’s lumen volume – always refer to the instructions provided with the specific short-term hemodialysis catheter kit being used.
A short-term hemodialysis catheter should be removed as soon as it is no longer clinically necessary. Prolonged dwell time beyond the intended short-term period exponentially increases infection and thrombosis risk.
Transition Planning: Nephrologists and vascular access coordinators should proactively plan for transition to a permanent fistula, graft or tunneled catheter when long-term renal replacement therapy is anticipated. Short-term devices are not substitutes for permanent access in patients with end-stage renal disease (ESRD).
Catheter Removal Technique: When removing the catheter, apply firm pressure to the exit site for at least 5 minutes to prevent hematoma formation. For femoral catheters or patients on anticoagulation, longer compression may be necessary. Document catheter tip integrity after removal.
Not all catheters are manufactured to the same standard. A reliable short-term hemodialysis catheter manufacturer produces devices with consistent wall thickness, kink-resistant materials, smooth-tip design and precise lumen dimensions – all of which affect blood flow adequacy and mechanical durability during dialysis sessions.
St. Stone Medicals Devices Pvt Ltd, is a recognized short-term hemodialysis catheter bulk supplier serving hospitals, dialysis centers and procurement teams across markets. When sourcing catheters in volume, procurement staff should verify that devices are biocompatibility-tested, CE-marked or FDA-cleared and comply with ISO standards applicable to intravascular catheters. Poor-quality devices with rough surfaces or weak hub connections directly increase the risk of thrombosis, hemolysis and air embolism.
Medical facilities should evaluate catheter performance data alongside price when selecting a short term hemodialysis catheter bulk supplier – because device failure during a dialysis session carries clinical and medico-legal consequences far exceeding the cost difference between products.
Preventing complications with a short-term hemodialysis catheter depends on consistent clinical practice across every stage – from correct site selection and sterile insertion to rigorous post-placement care and timely removal. Medical staff must treat every catheter placement as a high-risk procedure, not a routine one. Sourcing devices from a quality-assured short-term hemodialysis catheter manufacturer ensures the hardware supports safe clinical outcomes, but ultimately, it is staff training, protocol adherence and vigilant daily monitoring that determine whether complications are prevented or allowed to develop.

We are recognized as one of the leading manufacturers, exporters, and suppliers of a diverse range of high-quality medical devices in India. Our products, including Interventional Cardiology, Nephrology, Anaesthesia, General Consumables, and Critical Care Accessories, are designed to meet the highest standards of excellence, ensuring reliable performance and patient care.
A – 8, Kuraopokpi industrial Estate,
Thoubal (kakching) Imphal,
Manipur – 795001, India.